|
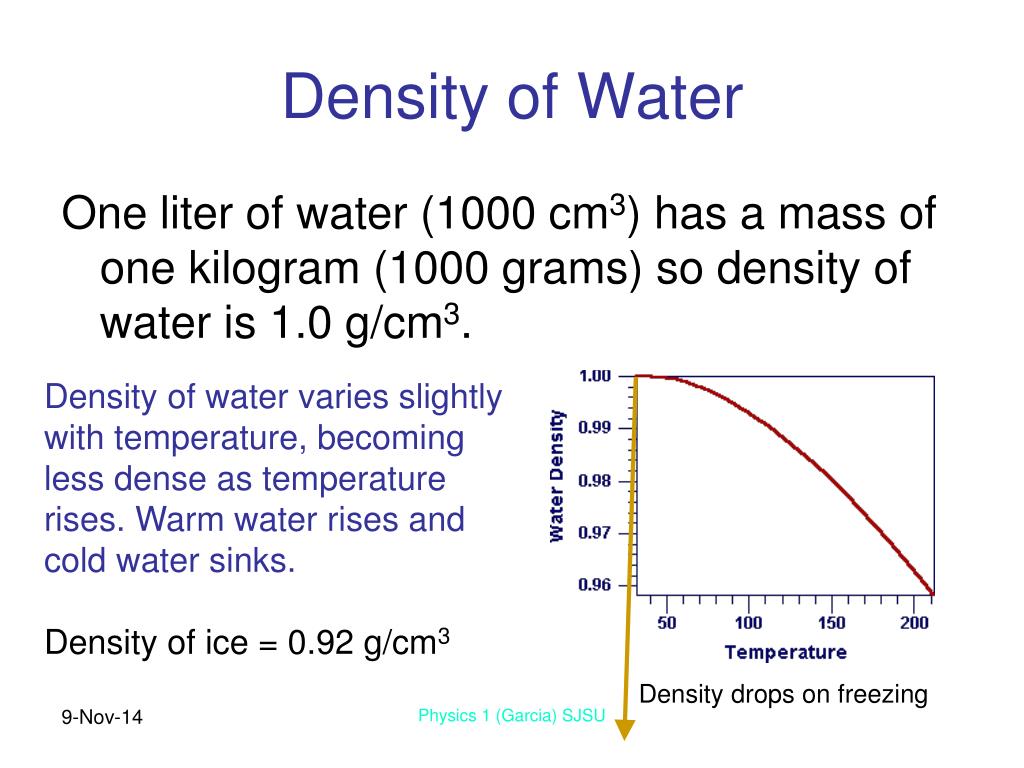
The question is based on Fig.1, which has a little pressure/temperature curve, but we don’t really need any of that. Question: Based on Fig.1, adding salt to water causes the boiling point of water to _. 
(Molarity is the concentration in moles per liter.) So if you have half a mole in a hypothetical 1 liter, then that’s 0.6 molar. Second, we converted the grams into moles of salt, and it came out to a little more than half a mole. First, taking that 1.028 kg/l and saying of that 1,028 grams, 3.5% of that is about 35 grams. So there were only two steps in the calculation. And you arrive at a number that’s a little more than half for the number of moles of salt in our hypothetical one liter. (As the denominator in a division problem gets smaller, the answer gets a little bit bigger). So this is a little more than half of a mole of salt in that liter of ocean water. So now you have 35 over 58, which is about 60. If you divide 35 grams by 58 g/mol, you’ll get the number of moles of salt in that liter.īut if you don’t want to do all that, 35 divided by 70 would be exactly a half. On the real test, you will have a periodic table that you can pull out to check the mass of sodium and chloride.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
